Examining the past with an eye on improving the future is something everyone does as a calendar year comes to a close. This month’s blog will honor that tradition, focusing on the future challenges and the past achievements of our industry.
One of the largest challenges our industry faced in 2012 was the economic impact expiring patents would have on some of our biggest pharma houses. As we braced ourselves for the fall off the patent cliff, we collectively held our breath and hoped that the decision makers would respond to this challenge by doubling down on R&D budgets and pipeline expansion.
When we review the events of 2012, it’s clear that the hopeful were not disappointed; the private sector innovated with a vengeance!
At year’s end 2011, we focused on the impending patent cliff and issued a blogthat discussed the relationship between patent protection and innovation. In the blog installment, we reviewed the original intent of laws granting patent protection – encouraging innovation while continuing to make affordable therapies available to the public simultaneously:
- By ensuring that innovative companies had the market protection they needed to recoup investments, earn a profit, and re-invest in research while also
- Ensuring that the protection granted to innovators expired in a reasonable amount of time so that other companies could quickly bring to market generic versions of those innovative products at the lowest possible price
There is no question that expiring patents presented 12 of our major drug makers very real financial challenges, and revenue reductions caused restructuring, layoffs, plant closures, and mergers. However, all the news was not bad.
- 2012 saw a 7.6% increase in the number of drugs in all phases of development including:
o 11.5% increase in drugs in pre-clinical phases
o an 8.8% increase in entities making it into phase III clinical trials
- The FDA approved 35 NMEs (New Molecular Entities) in 2012
- Of the 35 New Molecular Entities approved:
o 10 are treatments for cancer
o 1 is an HIV drug
o still other exciting NMEs will advance treatment of cystic fibrosis and multiple sclerosis
· Ongoing development activities include substantial scientific breakthroughs, including:
Not only will these discoveries provide our industry fuel for economic growth, they will also breathe life into start ups, expand the pipelines of the big companies, and provide the world’s population badly needed therapies.
Also noteworthy is the fact that 60% of new molecular entities approved were NOT developed by the top 20 pharmaceutical companies.
Many of the NMEs were developed by emerging pharma companies such as Organogenesis Inc., EUSA Pharma (Jazz Pharmaceuticals), and Vertex Pharmaceuticals. Smaller, leaner, and/or non-traditional research organizations that gained approval such as Incyte, BTG, and Duke University indicate that the face of research and development may be evolving in the face of these challenging financial times.
This is not necessarily bad news for big Pharma.
Many of these smaller organizations are equipped to devote all of their resources to research, and are more than happy to sell their pipelines to the big manufacturing houses in order to avoid the overhead that comes with development. It’s clear that diversity in the market place allows companies small and large, and/or old and new, to trim their design, and focus on the lifecycle stages that they are best suited to accommodate. All the while, being aided by FDA policies and programs designed to speed time to market, innovative and generic pharma and biologicproducts.
This seems like a win/win/win for large existing companies, small and emerging companies, and Joe Public. It seems that we are making great strides toward a more efficient path from discovery to market place.
2012 saw the FDA expedite the review and approval process via implementation of the fruits of their internal continuous improvement activities:
- Fast Track (Review): A process designed to speed up review of drugs developed to treat serious diseases and/or deliver solutions for previously unmet medical needs
- Accelerated Approval: A process applied to fast-tracked treatments, allowing the approval process to sustain the accelerated pace that the Fast Track review process set
- Priority Review: A program that prioritizes the resources FDA has to review applications, giving higher priority to pending applications that promise major advances in available treatments, or a new treatment for a space where no adequate therapy currently exists
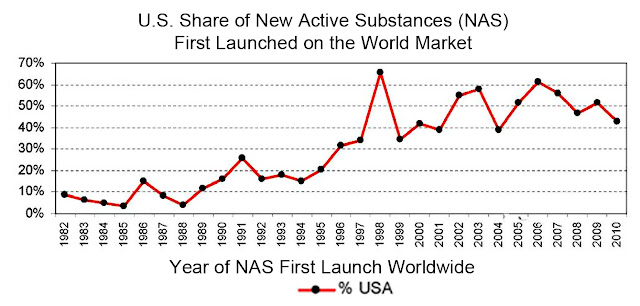
In 2012, FDA metrics for on time reviews were very encouraging, and indicate that these programs have had much success. The explosion of research, combined with the implementation of improvements in the licensing process, resulted in yet another year that saw the US lead the world in the introduction of new medical therapies, devices, and substances.
The graph below shows us that for the last 15 years, the majority of new medical substances worldwide have been delivered to the global marketplace by the US, and that 2012 saw a 15% increase over 2011.
2012 in Summary?
Big Pharma saw this coming and worried. They prepared, restructured, warned investors, and the patent cliff that loomed so large in 2011 came and went, and the output of US Pharma manufacturing continued to lead the world into a healthier future.
Smaller, leaner companies devoted to research also saw this coming, but from their perspective, they saw a market need and responded en force – discovery, innovation, and research efforts delivered volume and ground breaking diagnostics, treatments, and devices.
Big Pharma and small start ups decided to go the dance together, and newer, leaner options for pipeline development and sustenance arrived on the scene.
FDA also saw this coming, and spent the past few years evaluating their own processes, seeking opportunities for improvement. These improvements were implemented in time, and allowed FDA to keep pace with the rise in American innovation.
The real winner in 2012?
The population of the world.
The public in our country, and every other, saw generics where they were needed, and saw the approval of New Molecular Entities designed to treat and detect serious illnesses, including long rumored gene based cancer therapies.
We are Coda, and we wish all of our clients, and those they serve, a very happy and healthy holiday season. See you all next year!
© Coda Corp USA 2012. All rights reserved.
https://www.facebook.com/CodaCorpUSA
@Coda_Corp_USA
__________________________
Co-Authors:
Gina Guido-Redden and Corrine R. Knight
Coda Corp USA
866.872.2070
“Quality is never an accident; it is the result of high intention, sincere effort, intelligent direction and skillful execution. It is the wisest of many alternatives.”

 NAVIGATION
NAVIGATION