Leadership Insights
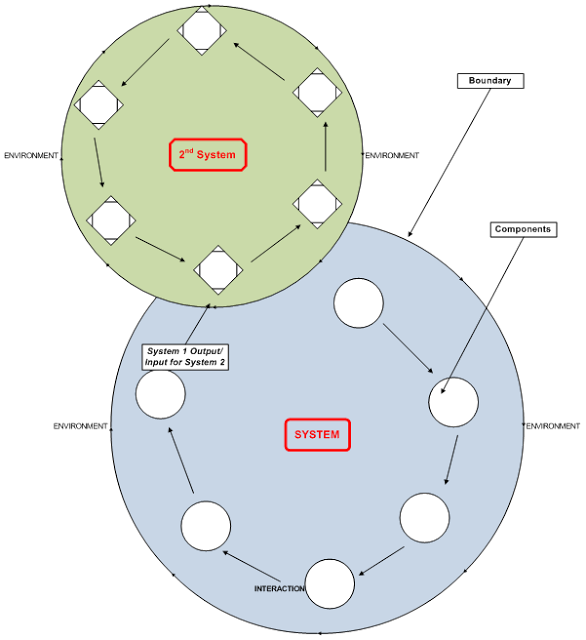
Think Systems: Cause Quality
What do all quality personnel have in common? We’re systems people. Or at least, we should be. Speaking in generalities, quality personnel create systems that have been designed to realize the definition of quality. The goal is always to define adaptive and effective systems that ensure our efforts cause quality to be realized and sustained. […]
Change Management and Change Leadership: A Critical Relationship
In this blog, I will evaluate the concepts of Management and Leadership within the most challenging environment – a changing environment. Let’s begin at the beginning, and distill the unique natures of Change Management and Change Leadership. Unique – But Complimentary The roles differ substantially, but they don’t compete with or inhibit each other; they […]
Defining Quality with Clarity
If you’re reading this, I’m assuming you earn your living in some sort of Quality Organization, but do you Speak Quality? And if you do, can you speak it fluently, or are you topped out at the conversational level? The reality is, if you’re like many people in Quality Organizations, you may not know. […]
Food and Drug Regulations; Too Many, Too Few, or Just Enough?
In light of the recent and well publicized drug shortages, the national debate on healthcare costs, and the slowing pace of new drug approvals, we felt the American consumer might appreciate a blog dedicated to the current relationship between drug manufacturers, the FDA, and Title 21. Federal Regulations and Consumer Protection: A Primer for the […]
Women’s History Blog: The Women of Science
Throughout history, women have had a significant impact on mathematics, science, and medicine. Despite the political, religious and social constraints of their time, these women sought the unattainable and questioned the unquestionable, often paying with their livelihoods or their lives. These remarkable trailblazers shattered myths, advanced our understanding of the universe, our own bodies and […]
The Difference Between?
Working in such a tightly regulated and hugely competitive industry and dealing with technologies that can at times be controversial, I expect to hear heated debates or energetic discussions regarding the nature and/or value of regulations, cutting edge technologies, or free market price wars. Because of the nature of our industry, I am never surprised […]
The One Voice Mission: A Quality Opus
I have worked within and around the life sciences community of Quality professionals for more years than I am ready to admit in a public post. Early on in my career, I listened avidly to every experienced person that I worked with, eager to take in everything anyone had to teach me. I was the […]
Understanding, Measuring, and Controlling the total Cost of Quality; The Holy Grail of The Modern Quality System
We decided this month to discuss the concepts, principles, and tools that aim to reduce operating costs while promoting and improving consumer-centric quality. Among this family of principles are those referred to as quality costing analysis models, or “Cost of Quality” (CoQ) models. Industry has been attempting to meet the challenges of measuring and controlling […]
Supplier Quality and the Total Cost of Poor Quality; Understand the Relationship and Avoid the Shortcuts!
During my two decade career, including a decade of consulting, I have integrated with a multitude of organizations, a wide variety of cultures, and a broad spectrum of projects, each with unique objectives, team members, and needs. This diversity of experience has been its own reward and continues to incentivize me to forego the obvious […]
Think Systems: Cause Quality
It is always appropriate to reflect on the definition of “Quality” and what that means for us. What is Quality? Some definitions were predictable, and straightforward, and many could be boiled down to generalities depending on the responder’s role in the supply chain. Consumers defined quality as having their functional and aesthetic expectations met for […]
Design and Validation – Drawing the Fine Line
Recently, several questions were submitted by readers, seeking clarity on the fine line between the stages of design and validation. While preparing individual answers to each question, we noted that dealing with them as a group presented an opportunity to address fundamental concepts comprehensively. This month’s blog is dedicated to highlighting the very real differences, as well […]
Be Better Than the Best
When I was in college, the robotics club held a fund raiser selling t-shirts that said “INNOVATION” – in all caps and BOLD lettering. I bought one – because I was so sure that someday, I would invent something that would change everything, for everyone. To date, I have not realized that dream. I […]
Women’s History Month: Names We All Need to Know
In honor of Women’s history month, I decided to write about the female pioneers in the sciences. These women blazed the trail that I and so many like me, would eventually follow. We have all walked in the mighty footsteps left by these amazing women; women who showed us all what could be achieved […]
Part 2: The Evolution of the Clinical Trial; Is Ethnopharmacology the New Frontier?
Last month, Part 1 of this series reviewed the history of some of the protections provided to the American consumer by Title 21 and the Food and Drug Administration. Last month’s installment focused on the development of the controls designed to evaluate the impact of utilizing our medicines in clinical situations; the history of Clinical […]
Part 1: The Evolution of the Clinical Trial – Is Ethnopharmacology the New Frontier?
Today’s American consumer is accustomed to the protections provided them by The Food and Drug Administration, protections that ensure the medicinal products we are exposed to are pure, safe, and effective. However, the current model of consumer protection applied to medicinal products has been, and continues to be, a product of evolution of knowledge, attained […]
Think Systems: Cause Quality
November, 2012 was celebrated as “World Quality Month.” ASQ CEO Paul Borawski asked this of industry: “What do you use as the best, most inclusive and illuminating definition of quality?” I followed the trail of replies as they posted, and it quickly became apparent to me just how diverse Mr. Borawski’s audience is, and how […]
The Biggest Story of 2012? Innovation!
Examining the past with an eye on improving the future is something everyone does as a calendar year comes to a close. This month’s blog will honor that tradition, focusing on the future challenges and the past achievements of our industry. One of the largest challenges our industry faced in 2012 was the economic impact […]
Cost Cutting and Quality
The last several years have been turbulent for the US pharmaceutical market. Unfavorable economic conditions have hit all sectors of the global economy. Historically, the pharmaceutical industry has been relatively immune to the downswings of the global economic cycle because illness, which provides the opportunities for these pharmaceutical companies to create and produce drugs, never […]
Big Pharma and The Blogosphere: Who’s Listening, and What’s Being Said?
If you’re reading this blog, it seems to suggest that you have an interest in user generated content that relates to the Pharmaceutical industry. If that’s true, you share that interest with me. I blog. I also read, monitor, and track articles daily and am, in every way, enamored by the blogosphere. Regularly reviewing industry […]
The Biosimilar Guidance Documents; Industry Responds
Regulatory Summary In an effort to incentivize the market place to introduce competitively priced biologics, the Biologics Price Competition and Innovation Act, passed by the Senate in 2009, was signed into Federal Law as part of the Patient Protection and Affordable Health Care Act by President Obama in 2010. Among other components the BPCI Act: […]
Quality Control: Overhead or Profit Center?
I have recently joined Coda’s team of Quality System bloggers. My professional background is rooted in the development and management of Quality Control Laboratories. The experience working to create, export, streamline, maintain, improve, and manage QC labs has not only been enjoyable, but has also given rise to many valuable insights and lessons. With that […]
Understanding, Measuring, and Controlling the total Cost of Quality; The Holy Grail of The Modern Quality System
As a follow up to our Lean article, we decided to continue the discussion of concepts, principles, and tools that aim to reduce operating costs while promoting and improving consumer-centric quality. Among this family of principles are those referred to as quality costing analysis models, or “Cost of Quality” (CoQ) models. Industry has been attempting […]
The USFDA Launches Initiative to Reduce Infusion Pump Safety Risks
The U.S. Food and Drug Administration has announced a new initiative to address what FDA’s Center for Devices and Radiological Health (CDRH) has referred to as “rampant” safety issues associated with the technology utilized by external infusion pumps. CDRH Director Jeffrey Shuren has called this a first for the agency, exercising its authority over an […]
Warning Letters: Roadmap to Corrective Action
Facilities should understand the importance of this “early-warning” message from FDA. Following an unsatisfactory facility inspection, the FDA has the ability to give individuals and firms an opportunity to take voluntary and prompt corrective action in response to deficient findings before it initiates an enforcement action. FDA uses the Warning Letter to achieve voluntary compliance […]
FDA’s Approach to Globalization
On Feb 4, 2010, Dr. Margaret Hamburg explained in detail the enormous problem facing the FDA. Globalization has made the task of monitoring imports and suppliers daunting and nearly impossible. The huge number of products imported, the vast number of foreign companies that create regulated products, and the increased complexity of the supply chain from […]
Enforcement Actions Rising
Recently, expanding global markets and increased volume of products has increased the real and present threat to consumers, as can be seen by recent public health issues; • lead based paint used by Chinese toy manufacturers, • deadly excipients in veterinary food products, • salmonella outbreaks in spinach and peanut products, and • Heparin contamination. […]
FDA TRENDS: WHAT TO EXPECT IN 2010
As the end of the year is approaching and plans are made for next year, it is important to be aware of the current strategy and budget of the FDA. The past five years have revealed a continuous decline in injunctions, warning letters and inspections from the FDA. The projections for 2010, however, clearly reveal […]
Talking the Talk
I subscribe to the fundamental belief system refered to as the GMPs. This is my belief system, the concepts are applicable in every aspect of life, and contribute value to every experience. I think some of those types of people think only about the actual wording of the regulations, and they never stop to see […]
A Graduation Note from a Seasoned Consultant
I have been a professional consultant for over a decade. Each project is different; some are small enough to complete independently, some require a team, and some require multiple teams and span the globe. Some contracts force you to expand your skill set, and teach you something practical, while some require teaching others what you […]

 NAVIGATION
NAVIGATION