If you’re reading this, I’m assuming you earn your living in some sort of Quality Organization, but do you Speak Quality? And if you do, can you speak it fluently, or are you topped out at the conversational level?
The reality is, if you’re like many people in Quality Organizations, you may not know. But if you find yourself explaining anecdotally, or relying on your organization’s acronyms to make your point, you may also find that you struggle to apply Quality concepts.
If so, I wrote this month’s blog for you.
This installment will focus on some critical fundamentals:
- The relationship between control and Quality
- The nature of Quality Control, within the larger construct of Quality Assurance
- The components of Quality Assurance within a larger Quality Management System
Begin at The Beginning: Quality is a Function of Control
Before we evaluate the relationship of Quality Assurance and Quality Control, let’s first remind ourselves, that the cGMPs fundamental principle is that Quality is a function of Control.
All of our Quality models are developed with this fundamental in mind.
Understand Quality
But what precisely is Quality? The long answer may depend on:
- Whether or not you are contributing to production of a product, or you are a consumer of a product
- The type of industry you work for (regulated or non-regulated)
- Your function within industry
In an earlier installment of the blog, we reviewed the results of the 2012 ASQ World Quality Month poll that queried members about their definitions. Not surprisingly, this is what they found:
- Consumers defined quality as having their functional and aesthetic expectations met for a low cost
- Retailers defined quality as having satisfied their consumers
- Engineers defined quality as increased efficiency and reduction of defects
- Manufactures defined quality as having produced defect-free product, on time, and for as high a profit margin as possible
- Sales people and marketers defined quality almost entirely in terms of customer perception
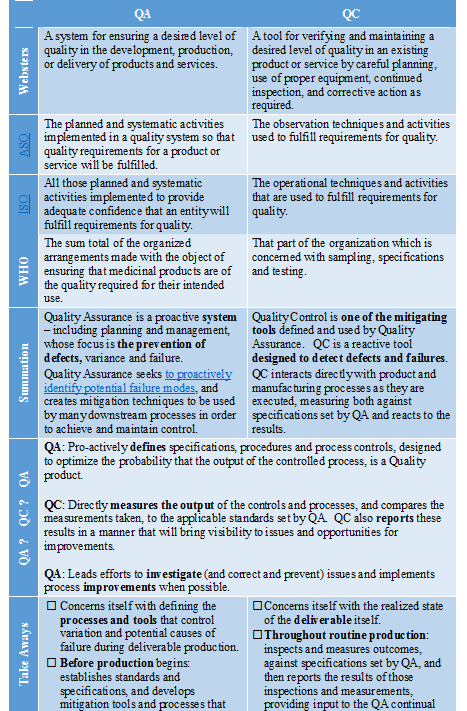
There are many published definitions of Quality Assurance, and of Quality Control. The table below reviews some of the most commonly referred to definitions:
- Monitor the routine manufacturing processes while they are being executed to confirm adherence to Critical Processing Parameters (CPPs).
- Measure the Critical Quality Attributes (CQAs) of the product to ensure that execution of operations processes resulted in product that met CQAs (for immediate use in lot release).
- Report the actual CPPs achieved in relation to the CQAs produced for each lot or device, both for product release and trending purposes (routine trending data feeds Continual Improvement efforts).
- Release of product if all CPPs have been adhered to and all CQAs have been met
- Documentation and investigation of processing deviations and quarantine of product, if CPPs have not been met.
- Documentation and investigation of product failure and rejection of product, if CQAs have not been met.
Managing and Maintaining: Quality Management and Quality Systems
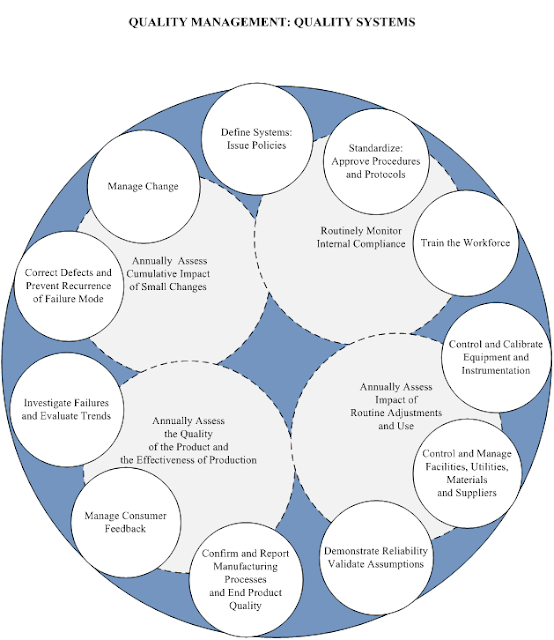
This maturation of the Quality Model (building systems, instead of just building product) is a realization that an understanding of how components affect each other cannot be developed by knowing only the components. We now recognize that there are some issues and assets that effect all stages of manufacturing including talent, knowledge, and tools, as illustrated in the following diagram.
- Placing the control systems in an outer concentric ring, as each occupies a space in time throughout the product lifecycle. Each control system is designed to mitigate an identified risk and, sequentially, increase the level of control by reducing the potential variance that we can expect when we routinely manufacture.
- Encompassing each of the control systems within a dotted line component; those components are regularly executed assessment tools, put in place to evaluate compliance with, and the effectiveness of, those control processes.
- Develop our understanding of the impact of our processes and the effects of our actions
- Address and utilize universal issues and assets consistently
- Identify trends and highlight relationships between upstream activities and downstream results
- Provide the most current, relevant, and objective information possible, allowing decision makers to make data-driven decisions geared toward the general goal of improvement
- Recognize that the supply chain is made up by multiple and equally critical perspectives, and facilitate consensus between them by introducing each to the system at the appropriate point in time
- Lean tools to reduce waste
- Six Sigma methodology to reduce variation
- Operational Excellence ideology to set goals
- Trending tools that promote managing with metrics
Summary
- Plan:
- Define the mission, vision, and goals (standards and specifications) to be achieved by an activity or a process.
- Identify the procedures, methods, and tools needed to achieve the goals (training, audits, supplier control, equipment and facility/utility control, validation, change management, in-process confirmation, in-process monitoring, final release testing, failure investigation, CAPA, and continuous improvement systems).
- Define the measures to be used to check the results of the process (procedures, methods, and acceptance criteria).
As always, we at Coda seek to provide Perspective with Purpose, in order to define Quality with Clarity!

 NAVIGATION
NAVIGATION